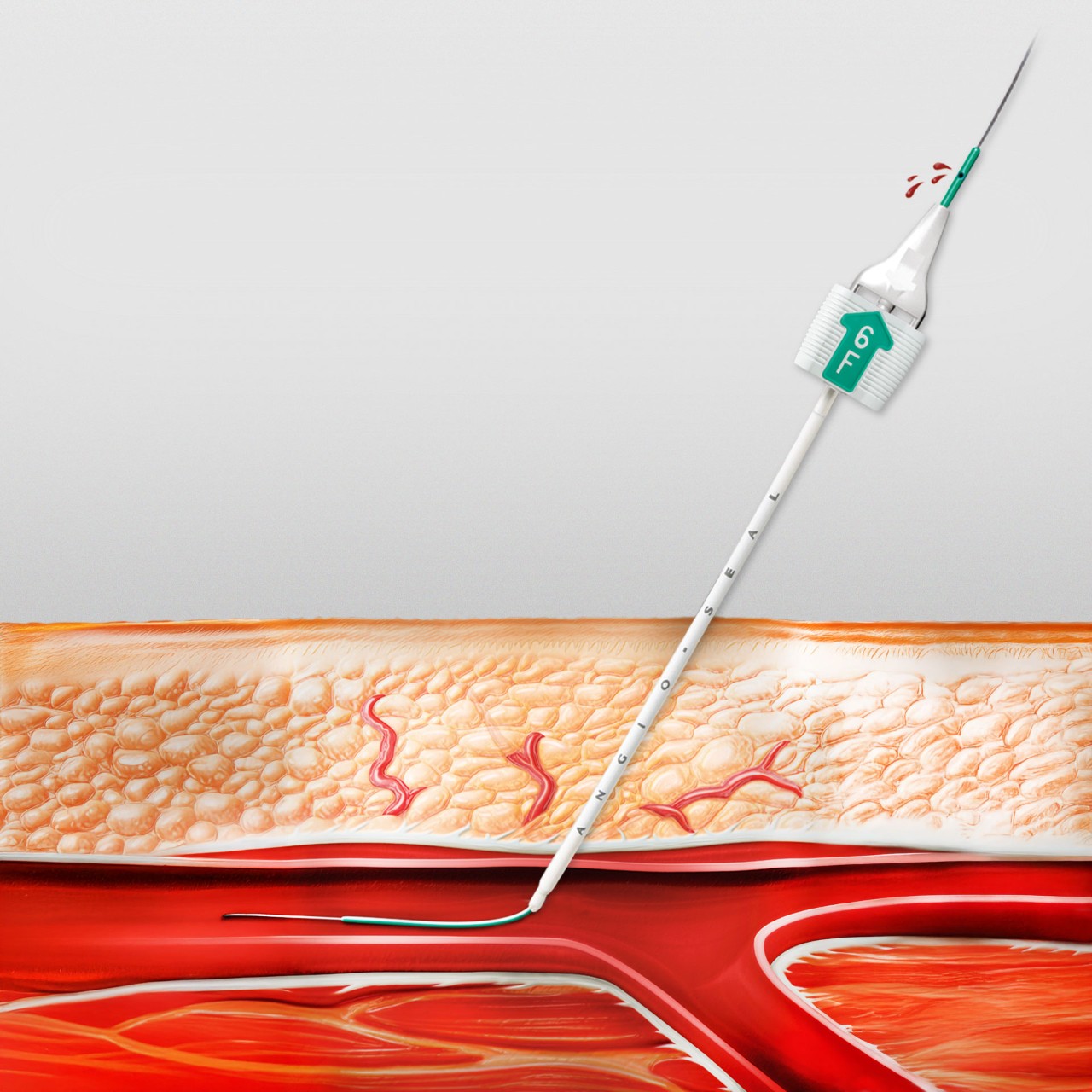
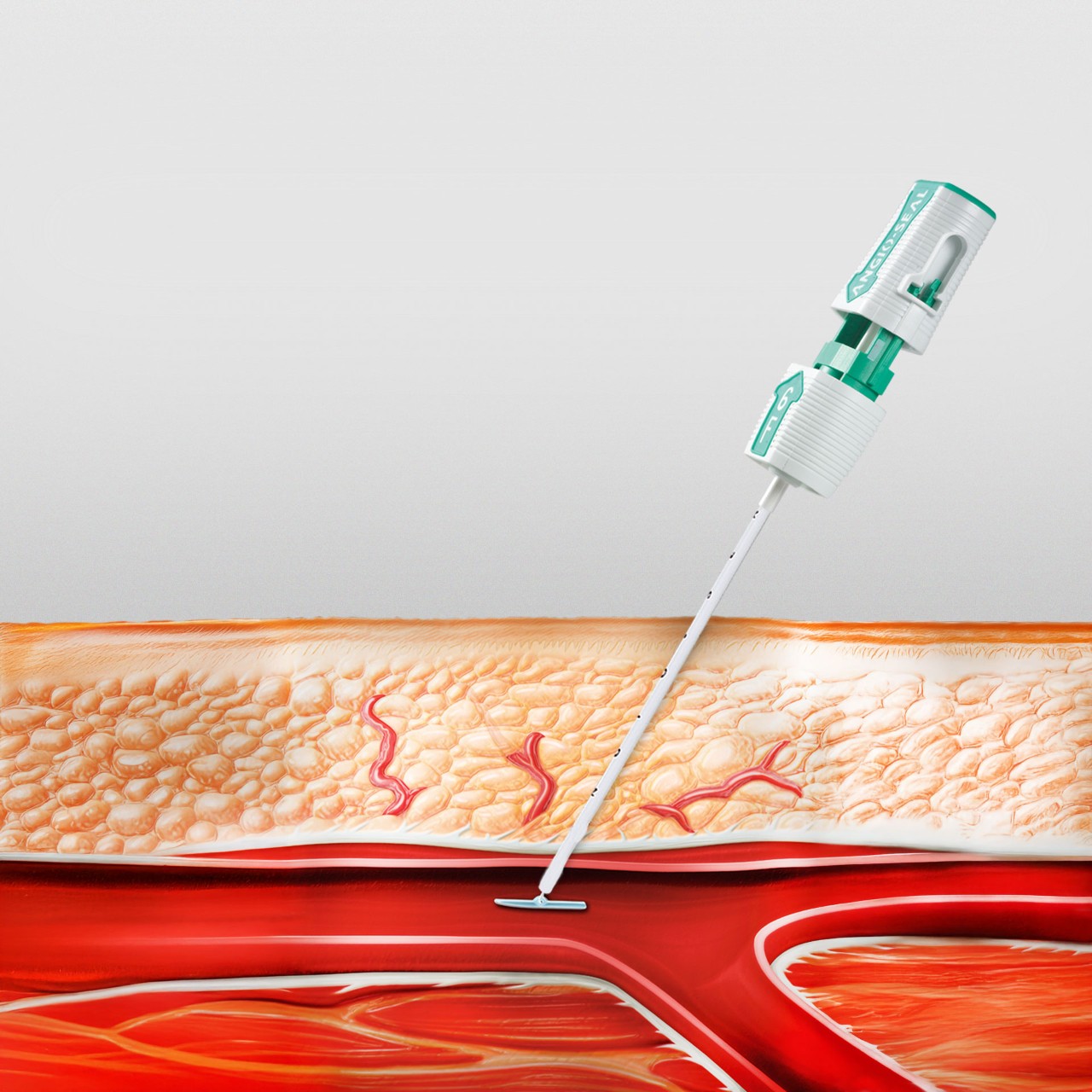
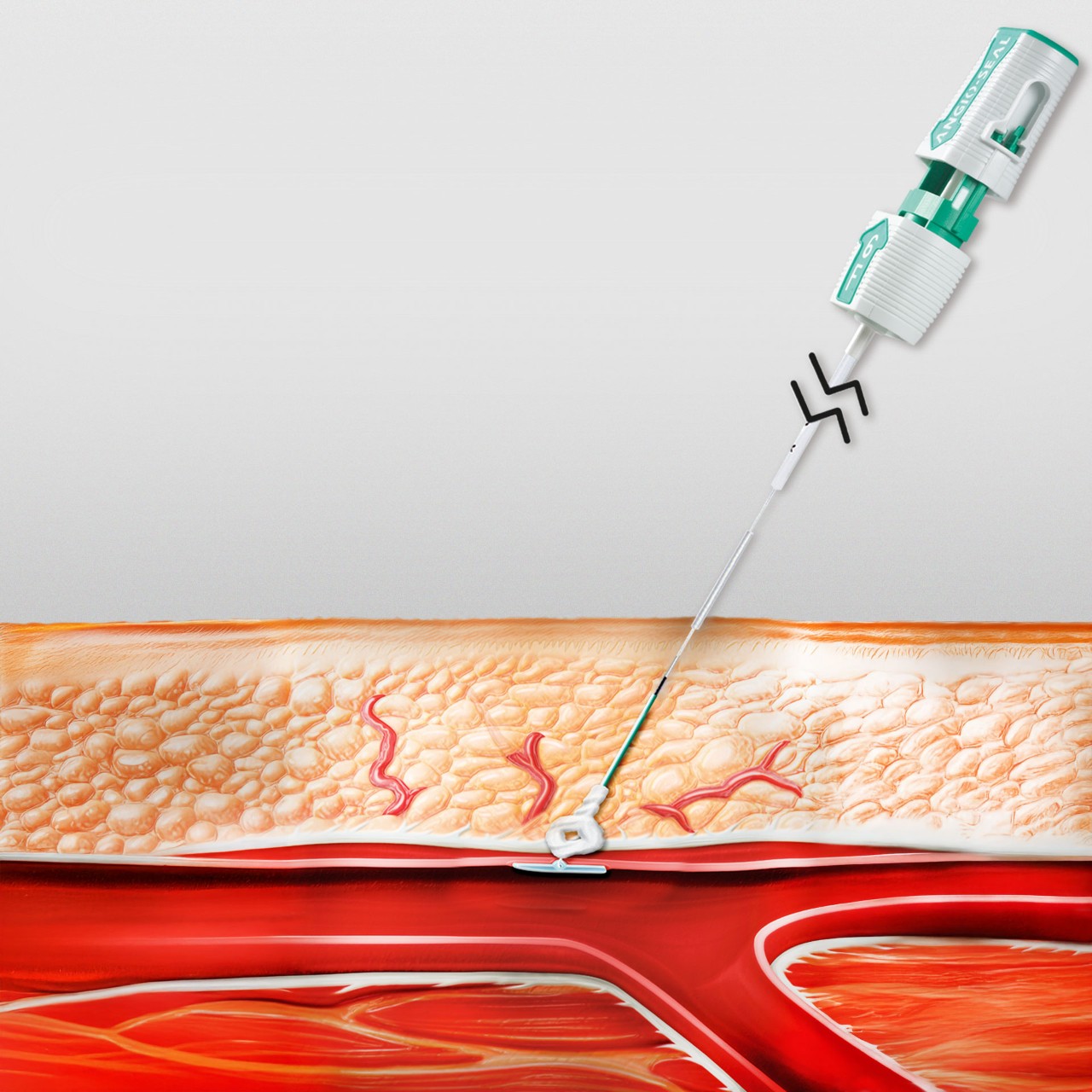
Successful Hemostasis
- The ANGIO-SEAL anchor creates a mechanical seal from the inside out and supports proper location for a reliable seal and collagen position1,2
- 99.7% deployment success3
- 97.8% hemostasis by device3
Bioabsorbable Components
All components are absorbed within 60-90 days1,2,4,5
1. Bioabsorbable Anchor: Begins to hydrate and soften 24-36 hours after deployment6 and is absorbed 95% at 42 days6
2. Bioabsorbable Collagen: Designed to conform to the arteriotomy for confident closure2
3. Bioabsorbable Suture: Tethers the anchor and collagen together, providing a secure seal2
Earliest Ambulation
- The only femoral closure device indicated for early ambulation for both diagnostic and interventional procedures9-15
Step-by-Step Instructions9
ANGIO-SEAL VIP Clinical Deployment Videos
Additional Resources
| Part Number | French Size | Guidewire Diameter (in) |
| 610130 | 6Fr | .035 Wire |
| 610131 | 8Fr | .038 Wire |
ANGIO-SEAL MRI Safety - Is ANGIO-SEAL MRI safe?
The implanted components of the device are MRI safe. The product is not made with natural rubber latex.9
Types of Vascular Closure Devices - What are some common types of vascular closure devices?
Percutaneous Closure Devices: These devices are inserted through a small incision in the skin and are guided to the puncture site to close the artery. They may use sutures, collagen plugs, or other mechanisms for closure.
Collagen Plugs or Sealants: Collagen-based plugs or sealants promote clot formation and provide a physical barrier to reduce bleeding, often used in combination with other closure methods.
Clip-Based Devices: These devices use clips or clamps to secure the arterial puncture site.
Vascular Compression Devices: These devices apply external pressure to the puncture site, aiding in hemostasis. They are commonly used alongside other closure devices.
References
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Kussmaul WG 3rd, Buchbinder M, Whitlow PL, et al. Rapid arterial hemostasis and decreased access site complications after cardiac catheterization and angioplasty: results of a randomized trial of a novel hemostatic device. J Am Col Cardiol. 1995;25(7):1685-92. 2.
- Nash JE, Evans DG. The Angio-SealTM hemostatic puncture closure device. Concepts and experimental results. Herz. 1999;24(8):597-606.
- Applegate RJ, Turi Z, Sachdev N, et al. The Angio-Seal Evolution Registry: outcomes of a novel automated Angio-Seal vascular closure device. J Invasive Cardiol. 2010;22(9):420-6
- Aker UT, Kensey KR, Heuser RR, Sandza JG, Kussmaul WG 3rd. Immediate arterial hemostasis after cardiac catheterization: initial experience with a new puncture closure device. Catheter Cardiovasc Diagn. 1994;31(3):228-32.
- Applegate RJ, Rankin KM, Little WC, Kahl FR, Kutcher MA. Restick following initial Angioseal use. Catheter Cardiovasc Interv. 2003;58(2):181-184.
- Tellez A, Cheng Y, Yi GH, et al. In vivo intravascular ultrasound analysis of the absorption rate of the Angio-SealTM vascular closure device in the porcine femoral artery. EuroIntervention. 2010;5(6):731-6.
- Kapadia SR, et al. The 6Fr Angio-Seal arterial closure device: Results from a multimember prospective registry. Am J Cardiol. 2001; 87:789-791.
- Manolis AS, et al. Simplified swift and safe vascular device deployment without a local arteriogram: Single center experience in 2074 consecutive patients. Indian Heart Journal. 2016; 68:529-538.
- Angio-Seal VIP Instructions for Use. ASIN0004. 2018-09-01 4
- StarClose SE IFU EL2101551 (10/2/14)
- MYNX Control Vascular Closure Device IFU 07/21
- MYNXGRIP Vascular Closure Device IFU 04/20
- VASCADE Vascular Closure System IFU 2611 Rev T, 11 AUG 2021
- Perclose ProStyle Suture-Mediated Closure and Repair System IFU EL2127594 (2020-11-03)
- CELT ACD Vascular Closure Device IFU-TS-004 Rev 4 (SCR 230)