Use With Confidence
- Only 0.021” wire on market and provides: greater rail strength to prevent buckling through scarred or calcified arteries1
Increase Efficiency
- The Glidesheath Slender Tibial Pedal needle technology requires less penetration force compared to competitive needle1
Reduce Complications
- Designed to minimize mechanical irritation to the artery1
Advance Your Approach to Tibial Pedal Access
Expand your options for procedural success with the proprietary technology of GLIDESHEATH SLENDER, designed for tibial pedal access.
Leverage Intricate Engineering
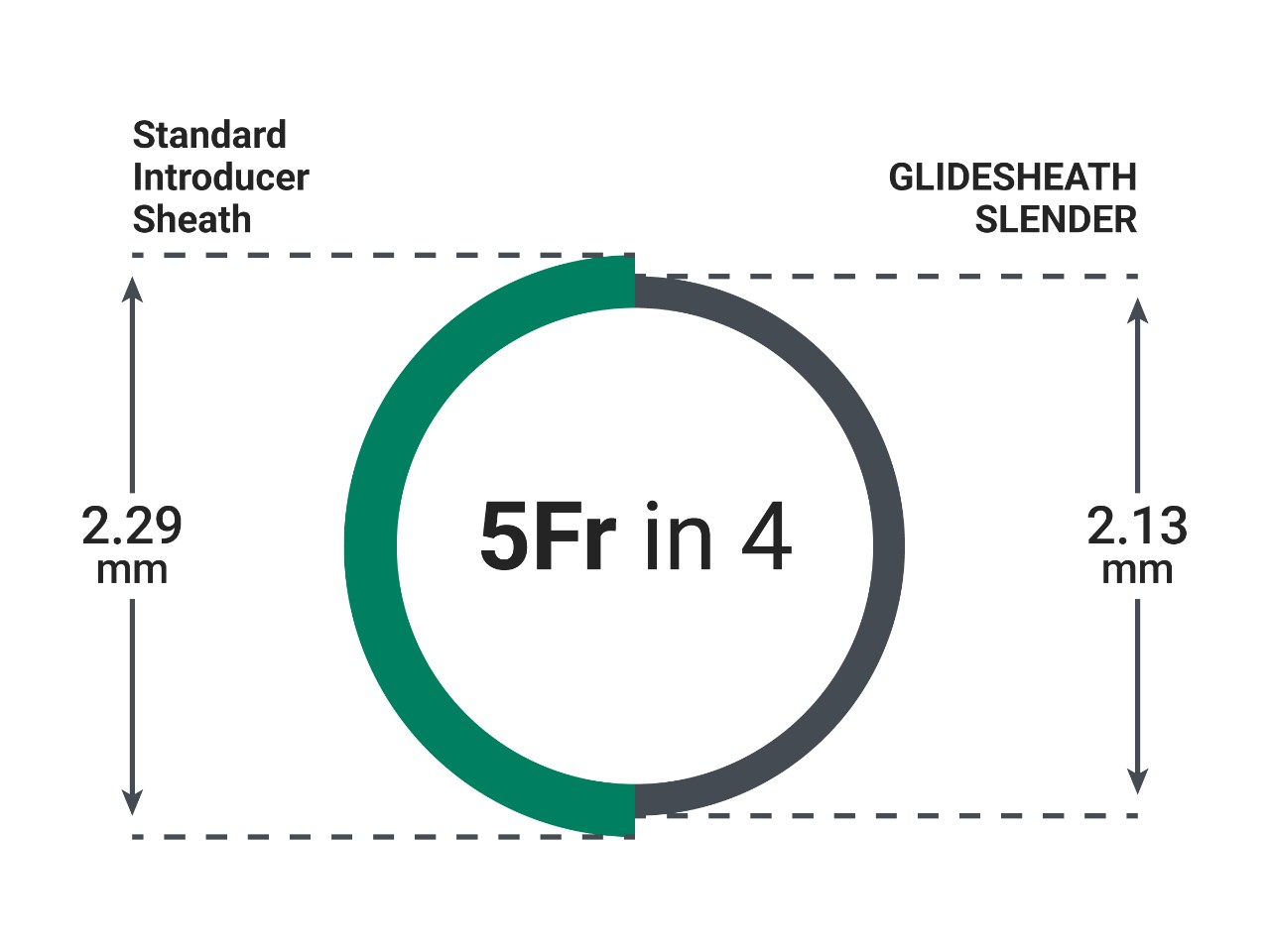
GLIDESHEATH SLENDER pedal access kit offers a thin-wall sheath option for procedures requiring a 5 Fr sheath.1

Proprietary Thin-Wall Technology
Proprietary thin-wall technology reduces the outside diameter of the introducer sheath by one Fr size while maintaining larger inner-diameter equivalent.
TERUMO Glide Technology™ hydrophilic coating enables easy insertion and removal.1
Access with Detailed and Sophisticated Design

Custom Guidewire
- An 0.021” wire provides greater rail strength to prevent buckling1
- Engineered with nitinol construction
Size: 0.021” | Length: 43 cm

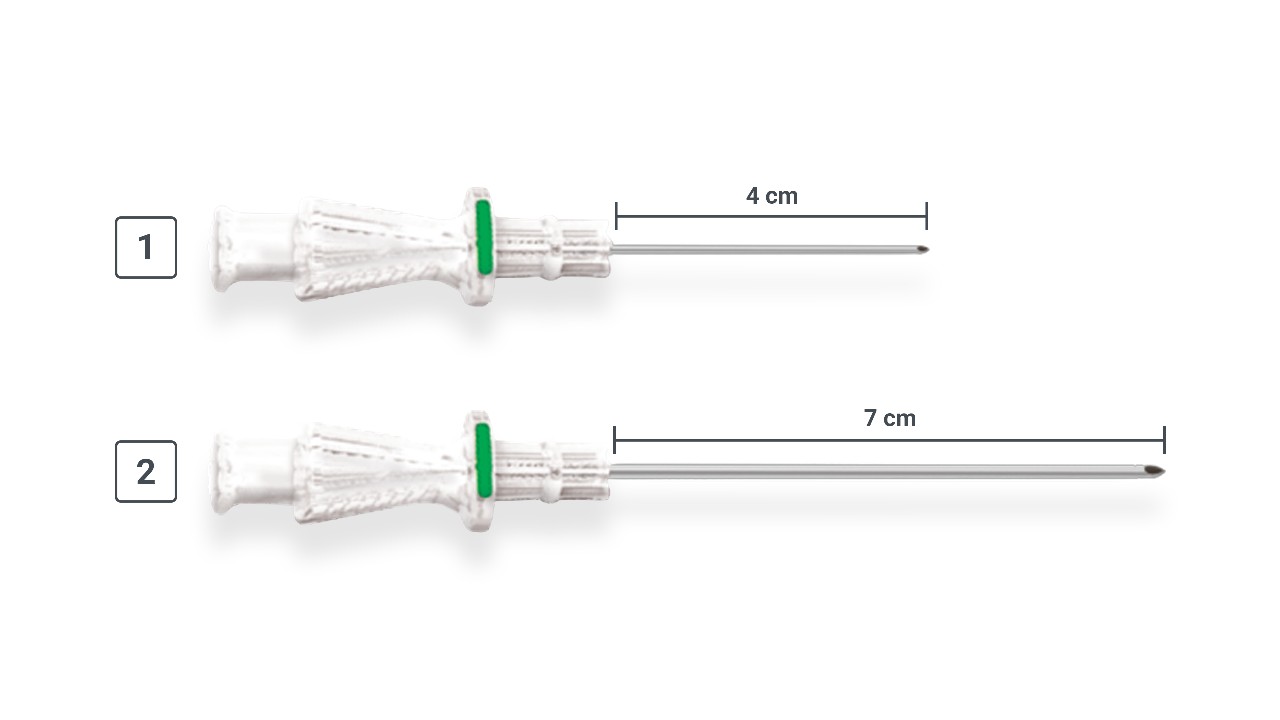
Custom Needle
- Tapered needle transitions from 21G tip to 19G shaft for better blood return
- Back bevel cuts help to facilitate a straighter entry1
- Echogenic tip for enhanced visibility
Length: 4 cm and 7 cm
Needle Size Comparison
Note: Needle diagram not to scale.
Additional Resources
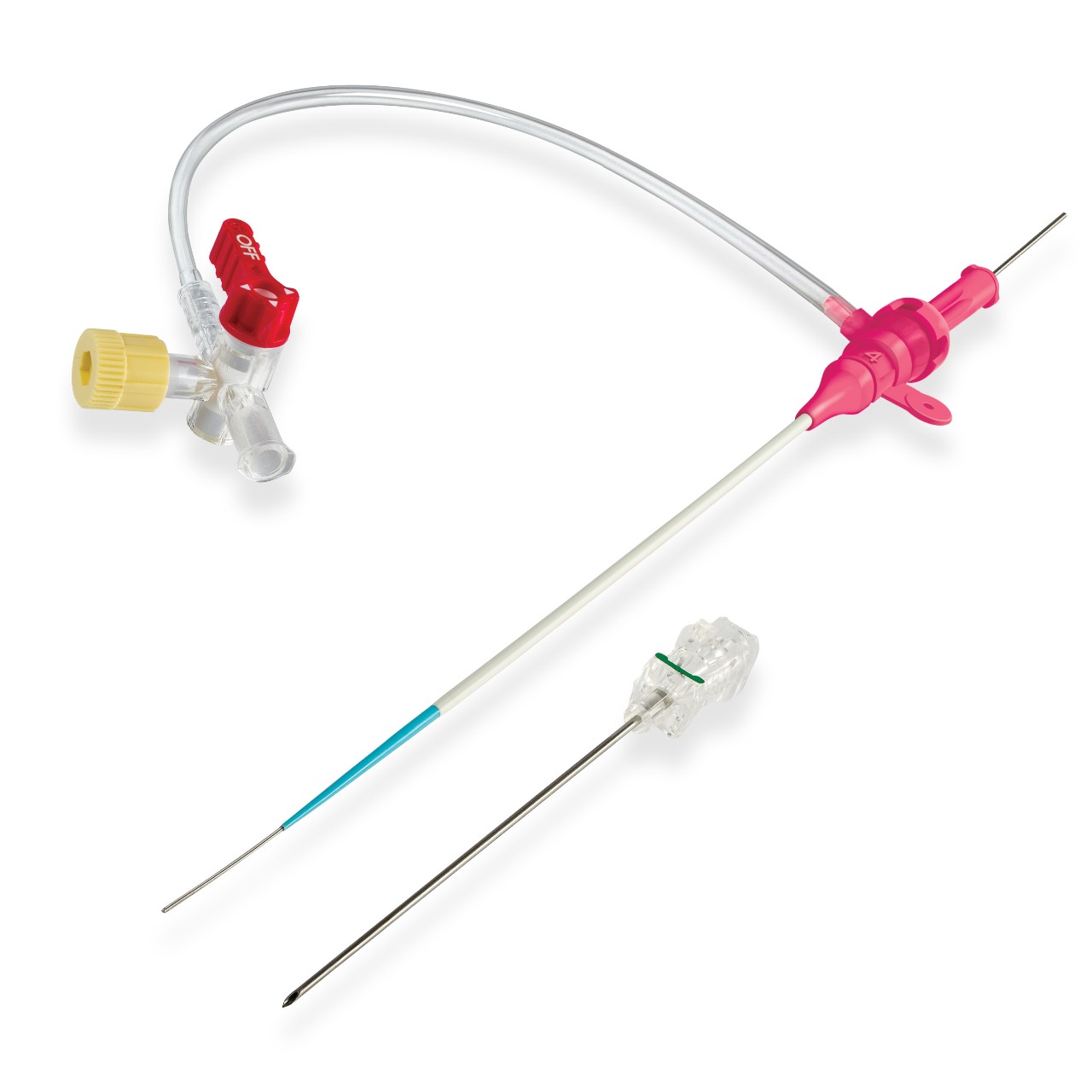
GLIDESHEATH SLENDER® Tibial Pedal Kit* includes:
| CODE | SHEATH | DILATOR | MINI-GUIDEWIRE | NEEDLE | NEEDLE LENGTH |
| 52-1050 | 5 Fr 10 cm | 0.021" | Nitinol/Floppy 0.021" x 43 cm |
21G-19G (Tapered) Echogenic |
4 cm (40 mm) |
| 51-1050 | 5 Fr 10 cm | 0.021" | Nitinol/Floppy 0.021" x 43 cm |
21G-19G (Tapered) Echogenic |
7 cm (70 mm) |
*5 per Shelf Box | Sheath Length: 10 cm | Wire Length: 43 cm | 4 cm or 7 cm Needle
What French sheath sizes are appropriate for tibial-pedal access in endovascular procedures, and how do pedal access kits impact outcomes?
Tibial-pedal access is critical in endovascular treatment of critical limb-threatening ischemia (CLTI), particularly when antegrade femoral access is limited. Given the small caliber of distal arteries (2–3 mm), 4 Fr and 5 Fr sheaths are most commonly used. 4 Fr sheaths are generally preferred due to their low outer diameter (~1.33 mm), which reduces the risk of arterial injury, spasm, and occlusion.1
Dedicated pedal access kits are designed to minimize trauma and improve procedural safety. Clinical studies, including the LIFE-BTK trial, support the use of low-profile access systems to improve technical success and reduce site-related complications.2
Related Products
References
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Data on file.
1 Mustapha, J. A. et al. (2019). J Endovasc Ther, 26(4), 524–532.
2 Conte, M. S. et al. (2022). J Vasc Surg, 76(1), 101–112.