Unique Formulation
- The first and only Polyethylene Glycol (PEG) microspheres. They are engineered for accurate embolization at the target area.1,2
Tight Calibration
- Available in a range of sizes from 75 μm to 1100 μm with a tighter size distribution*.
- This precise calibration may allow more predictable and targeted embolization 1,2**
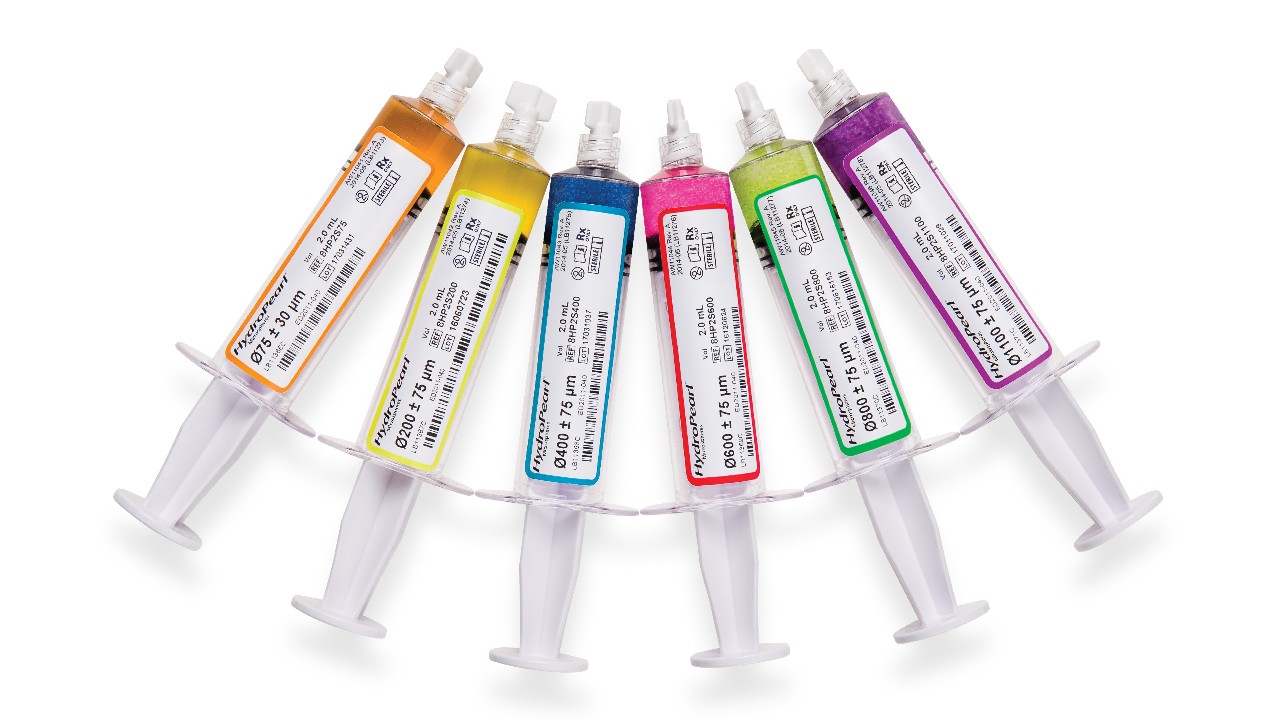
Available in Six Distinct, Color-Coded Sizes
- Distinct color coding reinforces your confidence in procedural success with the added assurance for proper syringe selection.
* 90% or higher of HYDROPEARL sizes are within specification as indicated on the labels.
** Calibration data is 6 lots per each size of randomly selected HydroPearl with 1 lot of competitive products. Sampling done at minimum 150 microspheres per syringe.
About HYDROPEARL Microspheres - Embolization Particles
HYDROPEARL microspheres are intended for the embolization of: 3,4
- Hypervascular tumors
- Symptomatic uterine fibroids
- Arteriovenous malformations (AVMs)
- Prostatic Arteries (PAE) for symptomatic benign prostatic hyperplasia (BPH)
HYDROPEARL Video – Suspension
HYDROPEARL Video – Robustness
HYDROPEARL Video – Calibration
In-Service Video
Additional Resources
HYDROPEARL Microspheres
| MICROSPHERE Sizes (μm) | Product Codes | Volume of Microspheres (ml) | Microsphere Color |
| 75 ± 30 μm | HP2S0075 | 2 | Orange |
| 200 ± 75 μm | HP2S0200 | 2 | Yellow |
| 400 ± 75 μm | HP2S0400 | 2 | Blue |
| 600 ± 75 μm | HP2S0600 | 2 | Red |
| 800 ± 75 μm | HP2S0800 | 2 | Green |
| 1100 ± 75 μm | HP2S1100 | 2 | Purple |
Terumo Catheter Compatibility1
| HYDROPEARL SIZES (μm) | PROGREAT® 2.0F | PROGREAT® 2.4F | PROGREAT® 2.7F | PROGREAT® 2.8F | GLIDECATH® 4.0F |
| 75 ± 30 μm | ✔ | ✔ | ✔ | ✔ | ✔ |
| 200 ± 75 μm | ✔ | ✔ | ✔ | ✔ | ✔ |
| 400 ± 75 μm | ✔ | ✔ | ✔ | ✔ | ✔ |
| 600 ± 75 μm | Non-Compatible | ✔ | ✔ | ✔ | ✔ |
| 800 ± 75 μm | Non-Compatible | Non-Compatible | ✔ | ✔ | ✔ |
| 1100 ± 75 μm | Non-Compatible | Non-Compatible | Non-Compatible | Non-Compatible | ✔ |
What are microspheres?
Microspheres are tiny spherical particles, typically 1–1000 micrometers(μm) in diameter, made from materials such as polymers, ceramics, or metals. Their small size and high surface‑area‑to‑volume ratio give them unique properties that make them valuable in medicine, pharmaceuticals, cosmetics, and various industrial applications. In healthcare, microspheres are often engineered for functions such as controlled drug delivery, diagnostic imaging, or targeted embolization, where they can block blood flow to specific tissues.
Terumo HYDROPEARL microspheres are a specialized medical example of this technology. They are the first and only embolization microspheres made from polyethylene glycol (PEG), a biocompatible and compressible material designed for precise, targeted vascular occlusion. Available in a tightly calibrated range of sizes from 75 µm to 1100 µm, HYDROPEARL microspheres offer predictable performance and can be selected based on clinical needs. They are intended for use in procedures treating hypervascular tumors, symptomatic uterine fibroids, arteriovenous malformations (AVMs), and prostatic artery embolization (PAE) for benign prostatic hyperplasia (BPH). Their consistent sizing and color‑coded system support accuracy and confidence during procedures.
Related Products
References
RX ONLY. Refer to the product labels and package insert for complete warnings, precautions, potential complications, and instructions for use.
- Data on file.
- Karolin J. Paprottka et al. In vitro study of physical properties of various embolization particles regarding morphology before, during and after catheter passage. Clinical Hemorheology and Microcirculation. 1386-0291/16.
- Per Instructions for Use PD111835 Rev. C Revised 2021-11
- HydroPearl 510(k) Letter. 22 Jan. 2020