Pushability
- Provides superior pushability to access and cross challenging lesionsa
Trackability
- Provides a superior ability of the guidewire to track and navigate the anatomy of the vasculature systema
Durability
- Provides superior shape retention for navigating small, tortuous vesselsa
Hybrid Wire for PAD/CLI
The addition of GLIDEWIRE ADVANTAGE TRACK™ Guidewire to your PAD/CLI toolkit provides you with superior performance1,a for your most challenging cases:
- Superior Pushability: High deformation resistance provides superior pushability to access and cross challenging above-the-knee (ATK) & below-the-knee (BTK) anatomy and lesions
- Superior Trackability: Provides superior ability to track and navigate anatomy of the vasculature
- Best-in-Class Durability: Provides superior shape retention for navigating small, tortuous vessels
- Superior Lubricity: TERUMO Glide Technology™ hydrophilic coatingb provides superior lubricity to support ease of progression through vessels
- Enhanced Visibility & Selectivity: Gold coil tips enhance visibility in conjunction with a 25 cm distal taper designed to minimize vessel trauma and improve selectivity of vessels
a When compared to Abbott Hi-Torque Command 18 LT and Boston Scientific V-18
b Referring to the distal 25 cm of GLIDEWIRE ADVANTAGE TRACK™ wire which features original GLIDEWIRE® construction
Product Specifications
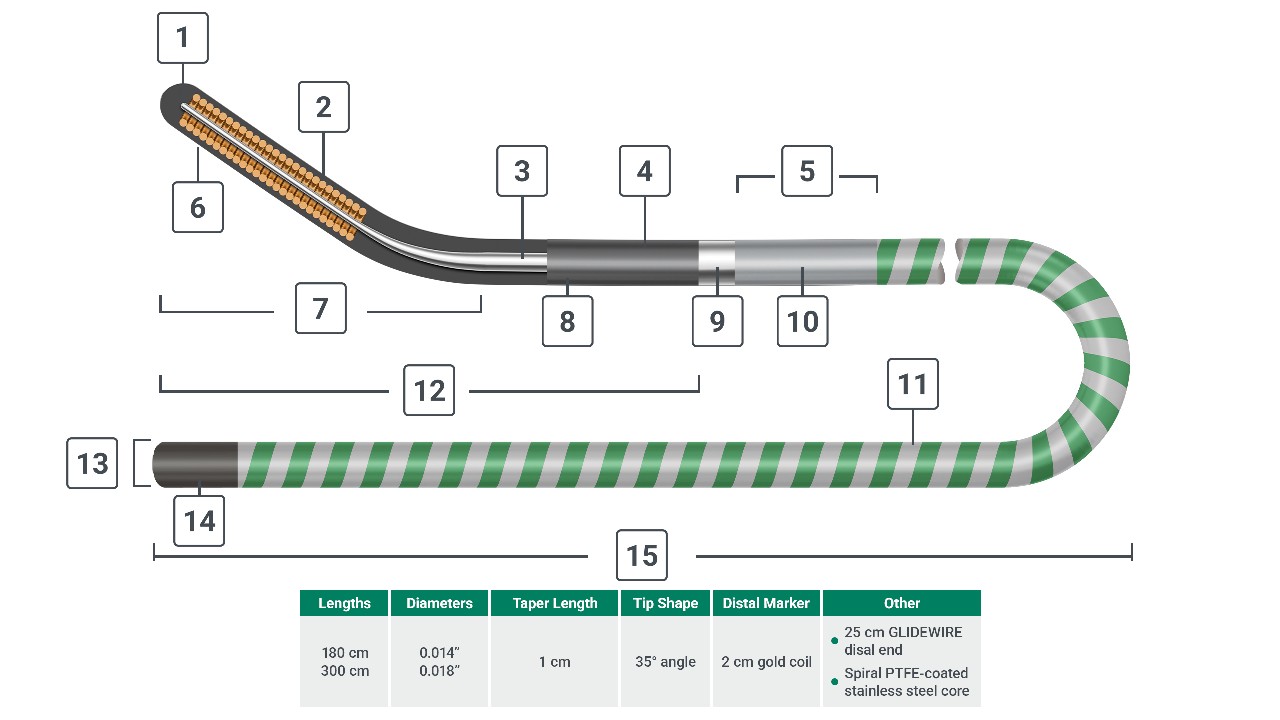
Nitinol core tip
2cm gold coil
TERUMO nitinol alloy core
TERUMO Glide Technology hydrophilic coating
Nitinol-Stainless Steel Connection
35° tip angle
Flexible tip length: 1cm
Polyurethane jacket with tungsten
Radiopaque band
Nitinol-Stainless Steel fusion joint
Stainless steel core with spiral PTFE coating
GLIDEWIRE construction (distal 25 cm)
Diameters: 0.014" & 0.018"
Uncoated proximal end: 2cm
Lengths: 180 cm, 300 cm
Additional Resources
GLIDEWIRE ADVANTAGE TRACK™ Peripheral Guidewire
| Product Code |
Diameter | Total Length | Distal Glidewire Length | Flexible Tip Length (Taper) |
Tip Shape |
| GAT1418 | 0.014" | 180 cm | 25 cm | 1 cm | 35˚ Angle |
| GAT1430 | 300 cm | ||||
| GAT1818 | 0.018" | 180 cm | |||
| GAT1830 | 300 cm |
Related Products
References
RX ONLY. Refer to the product labels and package insert for complete warnings, potential complications, and instructions for use.
1. Data on file.



